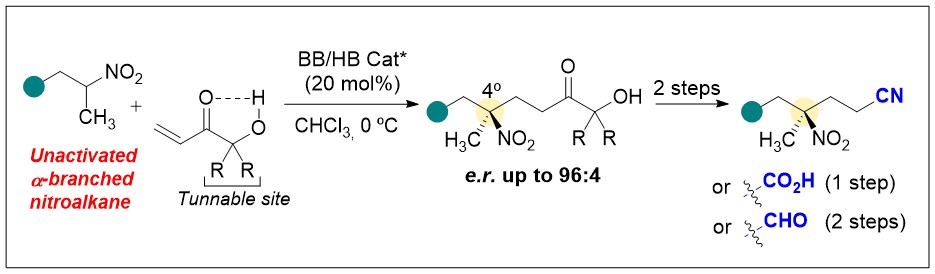
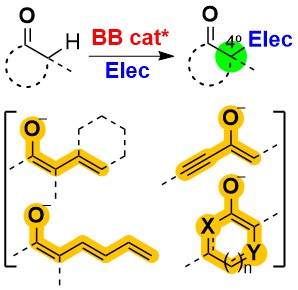
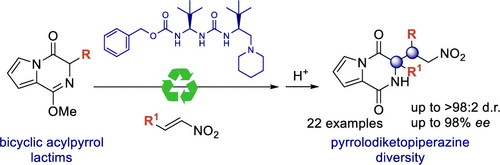
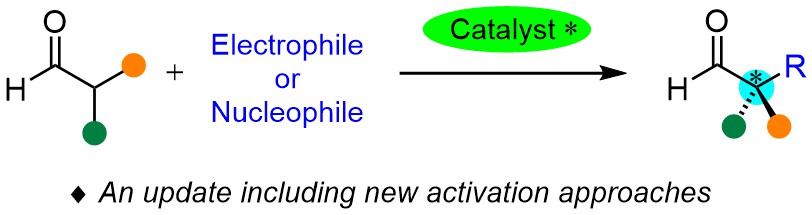
Publicaciones
—
10 Resultados por página
Publicaciones anteriores a 2010
- α-Hydroxy Enones as Achiral Templates for Lewis Acid-Catalyzed Enantioselective Diels-Alder Reactions J.Am. Chem. Soc, 2003; 125, 13942 - 13943
- Development of a new family of conformationally restricted peptides as potent nucleators of ß-turns. Design, synthesis, structure, and biological evaluation of a ß-lactam peptide analogue of melanostatin J. Am. Chem. Soc, 2003; 125, 16243 - 16260
- Design and Synthesis of a Novel Class of Sugar-Peptide Hybrids: C- Linked Glyco ß-Amino Acids Throuph a Stereoselective Acetate Mannich Reaction as The Key Strategic Element J. Am. Chem. Soc, 2002; 124, 8637 - 8643
- A Chiral Acrylate Equivalent for Metal-Free Diels-Alder Reaction: Endo-2-Acryloysoborneol J. Am. Chem. Soc, 2002; 124, 10288 - 10289
- Asymmetric Synthesis of ß–Mercapto Caboxylic Acid Derivatives by Intramoleculary. Sulfur Transfer in N-Enoyl Oxazolidine-2-thiones Promoted by Lewis Acids J. Am. Chem. Soc, 2001; 123, 5602 - 5603
- A ß-Lactam-Based Stereoselective Access to α,ß-Dihydroxy α-Amino Acid Derived Peptides with Either α,ß-Like or Unlike Configurations J. Org. Chem, 2001; 66, 4180 - 4186
- A strategy for the asymmetric aminohomologation of α,ß-dihydroxy aldehydes: Aplication to the synthesis of the southwest tripeptide segment of echinocandin B J. Org. Chem, 2000; 65, 41 - 46
- α-Oxy methyl ketone enolates for the asymmetric Mannich reaction: from acetylene and N-alcoxycarbonyl imines to ß-amino acids Angew. Chem. Int. Ed, 2000; 39, 1063 - 1065
- α-Oxy methyl ketone enolates for the asymmetric Mannich reaction: from acetylene and N-alcoxycarbonyl imines to ß-amino acids Angew. Chem. Int. Ed., 2000; 39, 1063 - 1065
- Hihly diastereoselective aldol reactions with camphor-based acetate enolate equivalents J. Org. Chem, 1999; 64, 8193 - 8200
—
10 Resultados por página